
All patients are given 5 g of idarucizumab as two intravenous boluses of 2.5 g, each administered over 5–10 min within 15 min of each other 6. This prospective study is enrolling two cohorts of dabigatran-treated patients: those with serious bleeding requiring reversal (group A) and those requiring urgent interventions that cannot be delayed for at least 8 h (group B). The efficacy and safety of idarucizumab are currently being evaluated in the phase 3 Study of the Reversal Effects of Idarucizumab on Active Dabigatran (REVERSE-AD number NCT02104947). In phase 2 studies in young or older volunteers with normal or moderately impaired renal function, idarucizumab rapidly reversed the anticoagulant effects of dabigatran in a concentration-dependent manner 5. Although the half-life of idarucizumab is prolonged in patients with renal impairment, the greater idaricuzimab exposure may be advantageous because these patients also have elevated plasma dabigatran levels. After intravenous infusion, the half-life of idarucizumab is about 45 min in subjects with normal renal function 5. 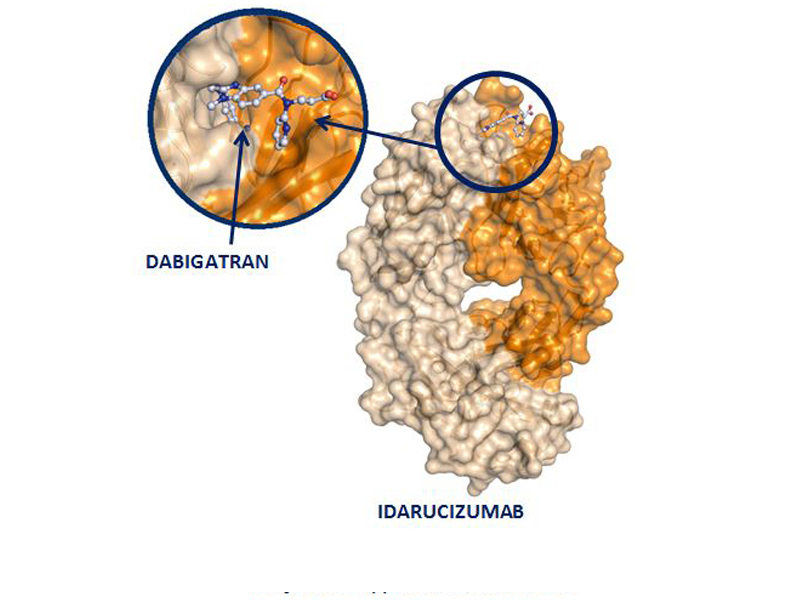
Idarucizumab and idarucizumab-dabigatran complexes are cleared by the kidneys, as is dabigatran. In addition to binding dabigatran, idarucizumab also binds the active glucuronide metabolites of dabigatran to form essentially irreversible 1:1 stoichiometric complexes 5. Idarucizumab is a humanized monoclonal antibody fragment that binds dabigatran with 350-fold higher affinity than that of dabigatran for thrombin. The properties of each are briefly reviewed. Comparison of the mechanism of action of the antidotesĮach of the three antidotes is a distinct chemical entity with a unique mechanism of action. This report (i) describes the mechanism of action of the antidotes, (ii) reviews the available clinical data, and (iii) provides guidance on potential indications for their use. 
Although each of these agents reverses the anticoagulant effects of the DOACs through different mechanisms and some are more specific than others, for simplicity, we will refer to all of them as antidotes. Ciraparantag (PER977), an agent reported to reverse the anticoagulant effects of all of the DOACs, is at an earlier stage of development. Andexanet alfa, the antidote for the oral factor Xa (FXa) inhibitors, is undergoing phase III investigation 4. 
Idarucizumab (Praxbind ®), the antidote for dabigatran, is now licensed in the United States and Europe. Three antidotes for the DOACs are under various stages of development. Nonetheless, antidotes for the DOACs would be useful as one component of strategies for management of serious bleeding, or for rapid reversal of the DOACs before urgent interventions. Although all anticoagulants can produce bleeding, the outcomes of major bleeds with DOACs are no worse than those with VKAs even in the absence of clinically available antidotes 3. As a class, the DOACs are at least as effective as vitamin K antagonists (VKAs) but are associated with less life-threatening bleeding, particularly intracranial hemorrhage 2. The direct oral anticoagulants (DOACs), which include dabigatran, rivaroxaban, apixaban, and edoxaban, are licensed for stroke prevention in patients with atrial fibrillation and for prevention and treatment of venous thromboembolism 1.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
